
Glomerular filtration rate (GFR) is a measure of how much blood is filtered by the kidneys each minute. It is the best overall indicator for how well an individual’s kidneys are functioning at a given moment.
Estimating GFR with cystatin C or creatinine
The gold standard for measuring GFR involves measuring plasma or urinary clearance of an exogeneous marker such as inulin, 99mTc-DPTA, 51Cr-EDTA, 125I-Iothalamate or Iohexol. This method involves a continuous intravenous infusion of the marker accompanied by repeat blood and/or urine collections at carefully timed intervals. Despite the accuracy offered by exogenous markers, measuring their clearance is too labour-intensive and time-consuming for use in ambulatory settings.
The preferred clinical alternative is to calculate an estimated GFR (eGFR) based on the serum or plasma concentration of an endogenous marker such as cystatin C or creatinine. The ideal endogenous marker is one that is produced at a constant rate in the body, is filtered by the glomerulus and not absorbed, metabolised, or secreted into the renal tract.
Timeline of events 1926 to 2020
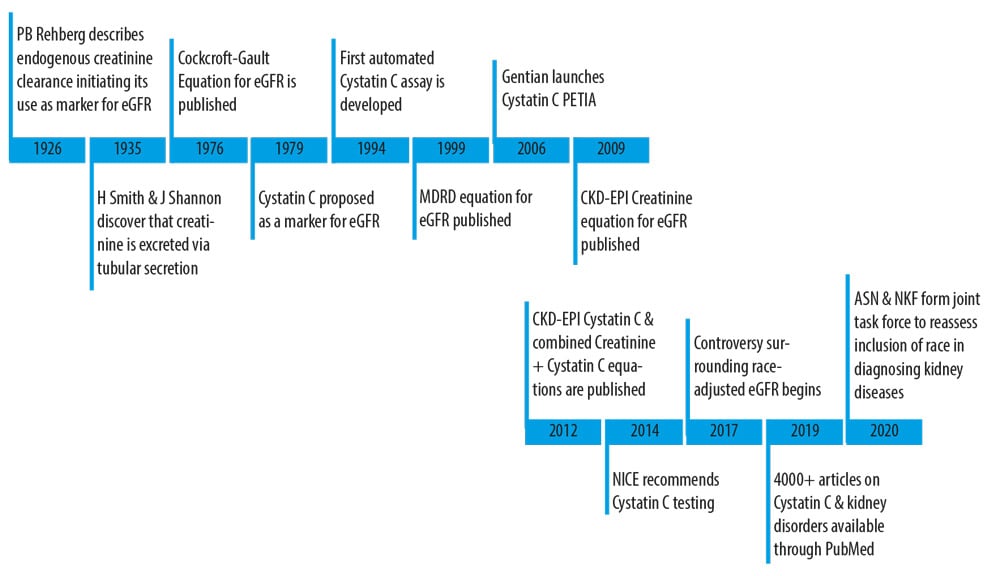
A search for the ideal endogenous marker of kidney function
1926
Paul Brandt Rehberg publishes “The rate of filtration and reabsorption in the human kidney” and in it describes the calculation of glomerular filtration rate through the exogenous administration of creatinine. Rehberg further concludes that tubular secretion of creatinine did not occur under normal physiological conditions1.
1935
James A. Shannon and Homer W. Smith publish “The excretion of inulin, xylose and urea by normal and phlorinized man,” noting a significant difference between inulin and creatinine clearance rates. Based on this finding, the pair conclude that creatinine is also excreted by tubular secretion and that GFR is better estimated by inulin clearance than creatinine clearance2,3.
1976
Donald W. Cockcroft and M. Henry Gault develop the first formula to predict creatinine clearance from serum creatinine in adult males. The formula was derived from observations made on a population of 249 male patients whose ages ranged from 18-92 years4.
1979
H. Löfberg and A. Grubb discover ϒ-trace (cystatin C) in human biological fluids. After observing high plasma concentrations of cystatin C in patients with advanced renal failure, Löfberg and Grubb posit that the protein must be catabolized in renal tubular cells following glomerular filtration5.
1994
The first automated particle-enhanced turbidimetric assay (PETIA) for cystatin C in serum and plasma is developed by Kyhse-Andersen6.
1999
The Modification of Diet in Renal Disease (MDRD) equation for predicting GFR from serum creatinine concentration is developed by Levey, A.S., et al. Researchers recommend routine use of the MDRD equation to predict GFR from serum creatinine concentrations citing more accurate estimates than those provided by creatinine clearance or the Cockcroft-Gault Formula. Of the 1,628 patients enrolled in the MDRD study, 88% were white7.
2006
Gentian Diagnostics AS develops the first platform-agnostic PETIA for quantification of cystatin C in human serum and plasma.
2009
The Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) established by the National Institutes of Diabetes, Digestive and Kidney Disease develops a new equation for estimating GFR from serum creatinine values. Researchers conclude that the CKD-EPI creatinine equation is “more accurate than the MDRD study equation across a vide variety of populations and clinical conditions.” One of the limitations noted in the study is the limited representation of racial and ethnic minorities across the 10 study populations (8,254 patients) used to develop the formula8.
2012
Inker, L.A., et al. publish the first equations that obviate race-reporting as part of eGFR calculation. The combined creatinine-cystatin C equation was found to perform better than equations that used either creatinine or cystatin c alone. The authors conclude that the combined equation may be “useful as a confirmatory test for chronic kidney disease"9.
2014
The National Institute for Health and Care Excellence (NICE) recommends using cystatin C to either confirm or rule out CKD in patients with a creatinine-based eGFR between 45 – 59 ml/min/1.73m2 that is maintained for at least 90 days without any other signs of kidney disease. The guideline was published in an attempt to reduce overdiagnosis of CKD stage 3a in the UK10,11.
2017
Beth Israel Deaconess Medical Center calls attention to the bias inherent in race-adjusted calculation of eGFR and becomes the first institution to report eGFR as a range between race-adjusted and race-unadjusted values12.
2019
Keyword search on PubMed for “Cystatin C” in combination of “Kidney Disorders” yields 4000+ academic publications
Dr. Anders Grubb highlights the importance of kidney biomarkers in laboratory medicine by mentioning that creatinine is the second most common analyte used in laboratory medicine; hemobglobin being the first13.
2020
The National Kidney Foundation and the American Society of Nephrology announce the formation of a “joint task force to examine the inclusion of race in the estimation of GFR and its implications for the diagnosis and subsequent management of patients with, or at risk for, kidney diseases14”.

Cystatin C in diagnosis and therapeutic assessment of renal function
Since its discovery in 19795, cystatin C has been widely recognised as a reliable marker for estimating glomerular filtration rate (GFR). GFR calculations performed using cystatin C are independent of protein intake, race, and muscle mass15-18. Equations that include cystatin C also predict GFR more accurately than serum creatinine in children*19, adults20, and older adults21. For these reasons, Clinical use of cystatin C has been recommended in guidelines published by Kidney Disease: Improving Global Outcomes KDIGO15 and NICE16.
GFR can be estimated with greater accuracy if both cystatin C and creatinine are used. However, cystatin C based equations are superior to creatinine-based equations in predicting end-stage renal disease, cardiovascular disease, hospitalisation, and death.
Clinical value of cystatin C:
- Avoids the creatinine blind area21
- Independent of muscle mass15-18
- Equations do not require a race adjustment15-18
- Improved dosing accuracy of drugs that are renally eliminated22
Contact us for more information about cystatin C
Do you want to know more about our Cystatin C Immunoassay or the clinical value of cystatin C? If yes, fill out the form below or send an email to marketing@gentian.com.
References
- Rehberg PB. Studies on Kidney Function: The Rate of Filtration and Reabsorption in the Human Kidney. Biochem J, 1926.
- Shannon JA and HW Smith. The Excretion of Inulin, Xylose and Urea by Normal and Phlorizinized Man. J Clin Invest, 1935.
- Shannon JA. The Renal Excretion of Creatinine in Man. J Clin Invest, 1935.
- Cockcroft DW and MH Gault. Prediction of creatinine clearance from serum creatinine. Nephron, 1976..
- Löfberg H and AO Grubb. Quantitation of gamma-trace in human biological fluids: indications for production in the central nervous system, in Scand J Clin Lab Invest, 1979.
- Kyhse-Andersen J et al. Serum cystatin C, determined by a rapid, automated particle-enhanced turbidimetric method, is a better marker than serum creatinine for glomerular filtration rate, in Clin Chem, 1994.
- Levey AS et al. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group, in Ann Intern Med, 1999.
- Levey AS et al. A new equation to estimate glomerular filtration rate, in Ann Intern Med, 2009.
- Inker LA et al. Estimating glomerular filtration rate from serum creatinine and cystatin C. N Engl J Med, 2012.
- Chronic kidney disease in adults: assessment and managment, N.I.f.H.a.C. Excellence, Editor. 2014: https://www.nice.org.uk/guidance/cg182.
- Lasserson DS et al. Requirement for cystatin C testing in chronic kidney disease: a retrospective population-based study. Br J Gen Pract, 2017.
- Kuehn BM. Medical Students Lead Effort to Remove Race from Kidney Function Estimates, in Kidney News. 2020, American Society of Nephrology: kidneynews.org. p. 1-3.
- AACC, The Use of Biomarkers in Chronic Kidney Disease and the Novel Disorder Shrunken Pore Syndrome. 2019: Youtube.com.
- Kimbrough J. Establishing a Task Force to Reassess the Inclusion of Race in Diagnosing Kidney Diseases, in A joint statement from the National Kidney Foundation and the American Society of Nephrology. 2020, National Kidney Foundation & American Society of Nephrology.
- KDIGO Guidelines 2012 published in Kidney International Supplements Vol3 Issue 1, Jan 2013.
- NCIC Clinical guideline (CG182) updated jan 2015 section 2.1 https://www.nice.org.uk/guidance/cg182/chapter/2-Implementation-getting-started.
-
Tangri N et al. Changes in dietary protein intake has no effect on serum cystatin C levels independent of the glomerular filtration rate, in Kidney Int 2011.
-
Grubb A, et al. eJIFCC 2017 Vol 28 NO4
-
Bjork J et al. Validation of standardized creatinine and cystatin C GFR estimating equations in a large multicentre European cohort of children, inPediatr Nephrol. 2019.
-
Lesly A et al. Estimating glomerular filtration rate from serum creatinine and cystatin C, in N Engl J Med 2012.
-
Schaeffner ES et al. Two novel equations to estimate kidney function in persons aged 70 years or older, in Ann Intern Med 2012.
-
Ebert N et al. Curr Opin Nephrol Hy. 2020.
*Not validated for children <14 years in the US
.png)

